 
An instance of a decomposition reaction can be:Ĭheck:- Empirical Formula Calculation Displacement reaction Basically, one reaction is converted into two similar reactions by breaking them out. This refers to the formation of a reaction made by dividing one reaction. 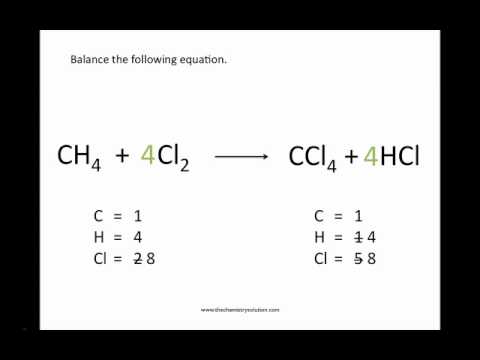
An example of a combination reaction can be:ĬaO (s) + H 2O (l) –> Ca (OH) 2 (aq). This refers to the reaction when two or more reactions of chemicals combine and form another one, then it is known as a combination reaction. There are 5 types of chemical reactions and are as follows: Combination reaction Then these are solved to balance the equation.įor balancing the unbalanced equation, the balancing chemical equations calculator is used to make it easier. These equations are known as unbalanced equations. Questions are given or the problems are not equal on both sides. The left side is considered a reactant and the right side is considered a product. The term unbalanced means that in an equation there are two sides, left and right. Algebraic equation balancing method– This refers to the unbalanced equations which include algebraic variables such as the stoichiometry coefficient.Traditional equation balancing method– It refers to the equation that is unbalanced and needs to be balanced in a traditional way.There are two types of balancing methods are as follows: So this kind of equation is used for solving complicated equations. It is evident from the above example that the reactants and products of an equation are balanced. The solution to this would be: CH 4 + 2O 2 → CO 2 + 2H 2O A question is given to balance the chemical equation: CH 4 + O 2 → CO 2 + H 2O 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
